Artist’s Profile of Erté[1]
Russian born artist – Romain de Trirtoff – better known as Erté (from the French pronunciation of his initials, R and T) – has produced a wealth of silkscreens that reflect his dazzling inventiveness, stunning elegance of wit and a refusal to produce formulated artworks.
Erté as a yound man.
He was born on the 23rd November 1892 in St. Petersburg (Russia) and died in Paris (France) on the 21st April 1990. In 1907, he lived one year in Paris. In 1910–12, he moved to Paris at the age of 18 to pursue a career as a designer. He made that decision despite strong objections from his father, who wanted him to continue the family tradition and become a naval officer. He assumed his pseudonym - Erté - to avoid disgracing the family name.
As a young boy he was fascinated by the Persian miniatures he found in his father's library. These miniatures were exotic, and brightly patterned designs, which continued to be important to him throughout his life since they influenced the development of his artistic style.
In 1915 he began a long relationship with Harper's Bazaar, during which time he created over 240 covers for the magazine. His fashion designs also appeared in many other publications, making him one of the most widely recognized artists of the 1920s.
The influence of his work as a result of his high visibility influenced an entire art movement that was to become known as “Art Deco”. He said around 1901 that: "…I did not discover Beardsley until I had already been in Paris for a year".
Throughout the 1920s and 1930s he also created original costume and fashion designs for many of the era’s most renowned screen actresses, including Joan Crawford, Lillian Gish, Marion Davies, Anna Pavlova, Norma Shearer and others. His creations for the stage included extravagant designs for productions at such venues as New York’s Radio City Music Hall, the Casino de Paris and the Paris Opera, as well as for the Folies-Bergères and George White’s Scandals.
At the age of 75, Erté was encouraged to embark on a new career and began to recreate the remarkable designs of his youth in bronze and serigraphy. The Art Deco movement was hence reborn. A lifetime of international success and recognition has ensured this unique artist's place in the annals of art history, and his original designs grace the permanent collections of prestigious museums throughout the world including New York’s Metropolitan Museum of Art, Museum of Modern Art, the Smithsonian Institution and London’s Victoria & Albert Museum.
Erté is perhaps best remembered for the gloriously extravagant costumes and stage sets that he designed for the Folies-Bergère in Paris and George White's Scandals in New York, which exploit to the full his taste for the exotic and romantic, and his appreciation of the sinuous and lyrical human figure.
As well as the music hall, Erté also designed for the opera and the traditional theatre, and spent a brief and not wholly satisfactory period in Hollywood in 1925, at the invitation of Louis B. Mayer, head of Metro-Goldwyn-Mayer.
After a period of relative obscurity in the 1940s and 1950s, Erté's characteristic style found a new and enthusiastic market in the 1960s, and the artist responded to renewed demand by creating a series of colorful lithographic prints (see below) and sculpture. The luxuriously illustrated artworks (shown below) contains a rich and representative selection of images, drawn from throughout Erté's long and extraordinary productive career.
In 1976 the French government awarded Erté the title of Officer of Arts and Letters, and in 1982 the Medaille de Vermeil de la Ville de Paris was bestowed upon him.
Some Lithographic Prints of Erté[2]
The Pursuit of Flore.
Oriental Tale.
Her Secret Admirers.
The Slave.
La Traviata.
At the Theatre: Trapeze.
At the Theatre: The Dancer.
At the Theatre: Golden Calf.
At the Theatre: Mélisande.
The Nile.
References:
[1] Erté Artist's Profile.
[2] New Erté Graphics, Erté, Dover publications, New York (1984).
Preamble
For your convenience I have listed below other post in this series:
Silk Designs of the 18th Century
Woven Textile Designs In Britain (1750 to 1763)
Woven Textile Designs in Britain (1764 to 1789)
Woven Textile Designs in Britain (1790 to 1825)
19th Century Silk Shawls from Spitalfields
Silk Designs of Joseph Dandridge
Silk Designs of James Leman
Silk Designs of Christopher Baudouin
Introduction
The period between 1764 to 1789 spanned the peak year (1764) for the export of English silks to the American colonies – the quantity of which was never surpassed. Eventually printed cotton became victorious and so the export decline of English silks became so drastic that it led to the demise of the silk industry in Britain.
The excellent essays of Natalie Rothstein’s about eighteenth century silks has resulted in two further major publications, namely, Barbara Johnson’s Album of Fashion and Fabrics (1987), and Silk Designs of the Eighteenth Century in the Collection of the Victoria and Albert Museum (1990).
The images and information contained in this post have been procured from a great book – The Victoria & Albert Museum Textile Collection, N, Rothstein, Canopy Books, Paris (1994). Her research on the collection is comprehensive and insightful. A “must have” for your ArtCloth library collection.
Today’s post will concentrate on the period from 1764 to 1789 in this on-going series.
Woven Textile Designs in Britain (1764 to 1789) [1]
The crisis of 1764-66 was compounded by several elements, namely:
(i) The public fashion tastes hit the silk industries all over Europe, eventually ensuring that printed cottons were considered by the public at large to be of high fashion, even though such opinions were not necessarily held by the elite.
(ii) The economic state of the London silk industry was such that it simply over produced and so the expensive price of silk products could not be sustained, since new markets could not be found for the product (lack of diversification) and old markets were shrinking.
Riots in Spitalfield.
A supply of good, cheap, raw silk could not be found, so that silks remained intrinsically expensive. The English industry blamed the import of French silks for the over supply. The unemployment amongst the English weavers was so bad, that a subscription list was published in order to assist them in these lean years. In the two years of unrest, demonstrations against their plight was peaceful, although Robert Carr, a well-known mercer had his windows smashed on suspicion that he was selling French silks and abuse was hurled at the John Russel (the Duke of Bedford) for remarks he had made about the weavers in the House of Lords.
John Russell - the fourth Duke of Bedford (painted by Sir Joshua Reynolds).
The London Gazette first came into print in 1665 and was originally called the Oxford Gazette - as it was decided that the King and his court should move to Oxford, along with the Gazette, in order to escape the plague. Once the plague dissipated, The Gazette returned to London. The copy (dated 1765) described the riots at Spitalfields as follows:
'Riots among the Spitalfields weavers, for many a century, were of frequent occurrence. The greatest riot was in 1765, when the weavers marched on parliament to protest against the defeat of a bill to tax imported silks.
They terrified the House of Lords into an adjournment, insulted several hostile members, and in the evening attacked Bedford House, and tried to pull down the walls, declaring that the duke had been bribed to make the treaty of Fontainebleau, which had brought French silks and poverty into the land.
The Riot Act was then read, and detachments of the Guards called out. The mob then fled, many being much hurt.'
The scale and organization of these popular protests frightened authorities. In 1763 the Journeymen issued their first list of "Prices" – wages to be accepted by both sides of the industry and in 1766 a law was passed making the formation of a trade union illegal. A period of bitter industrial dispute ensued.
The Spitalfields Act of 1773 onwards gave a half a century of industrial peace. A 'List of Prices' was agreed and 'new works' added as they became fashionable. The Spitalfields magistrates were given power to enforce them. The system worked until its repeal in 1826.
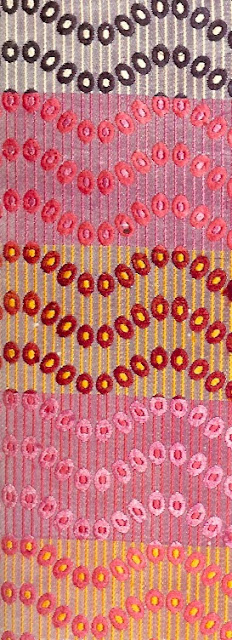
Silk Designs – 1764 to 1789
The quality of English silks remained excellent over the next few years (see image below).
Weaver’s sample, Spitalfields, ca. 1764.
Taffeta, brocaded in silver strip and thread, with a self-colored pattern in the ground.
Comment[1]: The sample is English and has an English inscription on the back, but has been inserted in a French order book.
Size: 14.25” x 21” (36.2 x 53.3 cm).
Courtesy of reference[1].
Manufacturers fell back on the formula they acquired from France; that is, a meander and garland facing one way, with a bunch of mixed roses and carnations facing the other. Increasingly a stripe was introduced as an additional motif, while the scale and size of patterns diminished. The less pattern there was, the cheaper it was to produce but this was offset by the decreased motivation that good artists had to design for the silk industry. There were plenty of pretty silk patterns as demonstrated by the four silk patterns displayed below.
Woven Silk, Spitalfields, ca. 1765-70.
Comment[1]: Tobine (cannel) ground, with green pattern wefts, brocaded with white silk and white silk fries.
Repeat Size: 3.5 x 1.75” (31.8 x 24.8 cm).
Courtesy of reference[1].
Nevertheless, if the market demanded large, original designs, then these would have been made. Below is a simple version of the meander and opposing sprig, and it is thus difficult to date.
Woven silk, Spitalfields ca. 1760-64.
Comment[1]: Tabby ground, with a white flushing warp.
Repeat Size: 7.5 x 4.67 “ (19 x 12.4 cm).
Courtesy of reference[1].
By 1770, the English silk industry had to adjust to a radical change in demand. There was now no market for furnishing silks - wallpapers, printed cottons and fine worsteds became much more important – and moreover, the market for dress silks was also changing. The modern softer, drape styles no longer required the heavy silks of the previous seventy years with their large-scale designs shown off on a sack-backed dress. The full length of a dress was no longer available to the designer since fashion now decreed deep flounces sometimes in contrasting materials. During the 1770s Neo-Classical taste began to affect both the patterns themselves and garments for which they were intended. The following two patterns from 1775 are typical.
Sample from silk weaver’s pattern book of Batchelor, Ham & Perigal, Splitalfield, ca. 1772 – 73.
Comment[1]: Satin and tobine (cannel) stripes with additional flush pattern.
Size: 6.75 (17.1 cm) high, together.
Courtesy of reference[1].
Woven silk, Spitalfields, ca. 1772-73.
Comment[1]: Tobine (cannel) ground, brocaded with colored silks and silk fries.
Repeat Size: 4.5 x 9.5” (11.4 x 24.1 cm).
Between 1770 and 1775 repeats were halved in length from 8 to 9 inches to as little as 3 or 4 inches in order to make woven silk more affordable. The two silk patterns below are typical of this design change.
Samples from silk weaver’s book of Batchelor, Ham & Perigal, Spitalfields, 1776, 1777.
Comment[1]: Brocaded taffeta.
size: 6” (15.2 cm) high.
Courtesy of reference[1].
Designers turned for inspiration to embroidery, printed cottons and the patterns of French silks (which were very similar in both style and coloring). The two silk patterns below from the pattern books of Batchelor, Ham and Perigal, supplement the evidence of surviving silks. Both demonstrate a leopard spot motif, which recurs until the early 19th Century, but is better carried out as a printed motif.
Woven silk, Spitalfields, ca. 1768-70.
Comment[1]: Tobine (cannel) ground with satin stripes, brocaded in colored silks, with additional flush pattern.
Repeat Size: 17 x 20” (38.1 x 48.9 cm).
Courtesy of reference[1].
Samples from silk weaver’s book of Batchelor, Ham & Perigal, Spitalfields, ca. 1772.
Size: 4.75” (12 cm) high.
Courtesy of reference[1].
The wriggle and spot of the silk pattern below, dated 1777, is another seen more effectively in the background of a printed cotton fabric. It was originally a pattern effect used in quilting. By contrast, even the ribbon, stripe and garland, which belong to the silk designers, were now used effectively by the calico-printers. The quality, which is particular to woven silk – its texture – was of no importance in a marketing sense for nearly all the samples and many of the surviving dresses were made from taffeta – for Spring – and satin – for the Winter season. The contrast in weave and types of thread, so important in previous centuries, was now abandoned.
Samples from silk weaver’s book of Batchelor, Ham & Perigal, Spitalfields, ca. 1777.
Comment[1]: Satin with a flush pattern.
Size: 4” (10.2 cm) high.
Courtesy of reference[1].
Within their limited field, the silks of the 1770s were nevertheless charming. Soft pinks contrasted with deeper chenille, delicate shades of green abound, with miniature Neo-Classical devices made by flush patterns.
Samples from silk weaver’s book of Batchelor, Ham & Perigal, Spitalfields, ca. 1776, 1777.
Comment[1]: Lightweight taffeta with a flush pattern made on a “monture” (i.e. draw loom).
Size: 2.5” (6.3 cm) high.
Courtesy of reference[1].
Samples from silk weaver’s book of Batchelor, Ham & Perigal, Spitalfields, ca. 1776, 1777.
Comment[1]: Brocaded Taffeta.
Size: 6” (15.2 cm) high.
Courtesy of reference[1].
The two above silk patterns are delightful but hardly exciting, except in a historical sense. The above silks were worn at the moment America was declaring its War of Independence, although from 1776 to 1783 hardly any silks were exported to America, which had been London’s major overseas customer. It was the merchant class on the eastern seaboard and their families who had bought fashionable silks. Many suffered politically in later years since they were loyalists. Even though the American market quickly recovered after the war, silks were no longer at the forefront of fashion and so the American market quickly downsized. Even though men’s silk waistcoats were becoming less expensive due to the fact that they shrank in scale and weight of decoration, fashionable men were now sporting embroidered waistcoats.
In the 1780s a marked decline in the use of flower designs in silk patterns became the order of the day. Stripes, zig-zags (on a minute scale compared with the 1750s), doodles, stars, and spots became the favorite motifs.
Samples from silk weaver’s book of Batchelor, Ham & Perigal, Spitalfields, ca. 1779.
Comment[1]: Satin. Zig-zags.
Size: 4.5” (11.4 cm) high.
Courtesy of reference[1].
Samples from silk weaver’s book of Batchelor, Ham & Perigal, Spitalfields, ca. 1781, 1782.
Comment[1]: Figured satin. Doodles.
Size: 2” (5 cm) high.
Courtesy of reference[1].
Samples from silk weaver’s book of Harvey, Ham & Perigal, Spitalfields, ca. 1786.
Comment[1]: Spots.
Size: 9.25” (23.5 cm) high.
Courtesy of reference[1].
In addition there were some extraordinary optical effects worthy of the 20th Century, while belt buckles sold well in the 1770s. Dark grounds for the "Winter Season" became increasingly fashionable from the late 1770s, as they were in chintz. The Maze & Steer pattern book of 1786 to about 1791 highlights silk patterns of that era.
Samples from silk weaver’s book of Harvey, Ham & Perigal, Spitalfields, ca. 1786.
Comment[1]: Satin with figured stripes.
Size: 5” (12.7 cm) high.
Courtesy of reference[1].
Samples from silk weaver’s book intended for waistcoats. Maze & Steer, ca. 1786, 1787.
Comment[1]: Figured satins.
Size: 3” (7.6 cm) high.
Courtesy of reference[1].
The above silk patterns were fundamentally embroidered designs adapted for another medium – for it was in these years that the Lyon Chambre de Commerce was bitterly complaining about the unfair competition from embroiderers. The vast majority of waistcoats surviving from the 1780s and 1790s were embroidered and whilst the silk waist coat patterns shown above are fair, they cannot compare with the splendid French embroidered rivals.
When Eden’s Free Trade Treaty was being negotiated with France in 1786, the silk industry in London was sufficiently strong and vigorous to secure exemption from its provisions, so that French silks remained excluded from England and vice versa.
The first attempts to set up a gauze industry in Paisley were apparently made by Spitalfields masters in the late 1780s and 1790s.
Reference:
[1] The Victoria & Albert Museum Textile Collection, N, Rothstein, Canopy Books, Paris (1994).
Preamble
On this blog spot there are posts that center on my “Wearable Art” (e.g. scarves, digital or analogue created fabric lengths etc.) For your convenience I have listed these posts below.
A Selection of My Scarves
Leaves Transformed: A New Collection of My Digitally Designed Fabrics
My New Silk Rayon Velvet Scarves@Purple Noon Art And Sculpture Gallery
My Fabric Lengths@QSDS
My Fabric Collection:"Oh, Oh Marilyn and Mona!"@Spoonflower
2013 Australian Craft Awards – Finalist
My Scarves@2014 Scarf Festival: "Urban Artscape" Pashminas
My New Scarves and Fabric Lengths
New Range of Silk Neckties - Karma and Akash
AIVA: My New Hand Dyed and Hand Printed Fabric Design
New Colorways For My 'Cultural Graffiti' Fabrics
Byzantine Glow: A New Collection of My Digitally Designed Fabrics
Wall Flower: A New Collection of My Digitally Designed Fabrics
Ink Fern - A New Collection of My Digitally Designed Fabrics
Celebratory Fireworks
My New Silk ArtCloth Scarves
New ‘Unique State’ Silk ArtCloth Scarves
UBIRR - My New Hand Dyed & Printed Fabric Design
Renaissance Man - My New Hand Dyed & Printed Fabric Design
Banksia - My New Hand Dyed and Hand Printed Fabric Design
Ginkgo Love - My New Hand Dyed and Hand Printed Fabric Design
Garden Delights I & II - My New Hand Dyed and Hand Printed Fabric Design
Wallflower III - My New Hand Dyed and Hand Printed Fabric Design
Rainforest Beauty - Collection My New Hand Dyed and Hand Printed Fabric Design
Spring & Autumn Flurry Collection - My New Hand Dyed and Hand Printed Fabric Design
La Volute Collection - My New Hand Dyed and Hand Printed Fabric Design
Urban Butterfly - My New Hand Printed Fabric Design
Acanthus Dream - My New Hand Printed Fabric Design
“Cascading Acanthus” - My New Hand Dyed and Hand Printed Fabric Design
My New Hand Dyed and Hand Printed 'Rainforest Beauty' Pashmina Wraps Collection
My ArtCloth Tea Towels: A New Collection of Digitally Designed Products
Through the Land it Roared . . . ArtCloth Shawl
My New Hand Dyed and Hand Printed ‘Urban Codes - Series 1’ Collection
Urban Moonlight - My Post Graffiti Doily
My New Hand Printed Fabric Design - "Morocco" ArtCloth
‘Vine Glow’
“Bush Banksia’s” Collection"
Releasing My New - ‘Unique State’ ArtCloth Scarves
‘LRSP’ A New Collection of Digitally Designed ArtCloth Textiles
If you like any of my artworks in the above links, please email me at - Marie-Therese - for pricing and for any other enquiries.
A Brief Timeline of Scarves[1]
The roots of the origin of the English word "Scarf" come from the Old French word "escreppe", which translates as a purse suspended around the neck. Nowadays the word scarf indicates any neckerchief, especially those worn for warmth. The Oxford Dictionary defines the word "Scarf" as a length or square of fabric worn around the neck or head: for example, "She tucked her woolly scarf around her neck."
Scarves have a long history, but not all of it has been well documented. In fact one website[1] has given a brief timeline of its history, which we have reproduced below and which now appears on our post of Timelines of Fabrics, Dyes and Other Stuff.
1350 BC: Egyptian Queen Nefertiti wore a finely woven scarf topped by a conical headdress.
1000 BC: Chinese sculptures featured scarf-like, fringed rectangular pieces of cloth.
230 BC: Warriors of the Chinese Emperor Cheng wore scarves made of cloth, which marked military rank.
10 AD: Romans wore a linen kerchief or “sudarium” (Latin for “sweat cloth”) knotted around the waist or around the neck.
60 AD: The Emperor Nero rarely appeared in public without a sudarium around his neck.
ca. 1150: Eleanor of Aquitaine wore scarves in a “gossamer cascade” from the tip of a tall pointed hat, starting a noted fashion trend of the Middle Ages.
1261: Egyptians adopted a dance style known today as belly dancing. Costumes included a scarf-like belt worn low on the hips.
1600: Croatian mercenaries wore scarves to signify rank.
1783: The Third Duke of Krakow was said to have invented the knitted scarf in this year.
1786: Napoleon Bonaparte sent his first wife Joséphine de Beauharnais cashmere scarves from India during his travels.
1810: Renowned composer Beethoven fell in love with Therese Malfatti and tried to win her heart by adopting a new look including fashionable suits, shirts and silk neck scarves.
1837: Hermès, French ready-to-wear retailer famous for its graphic silk scarves, was born.
1837: Queen Victoria came to the throne and popularizes fanciful accessories such as scarves. In the Victorian era in particular, these accessories aided to differentiate between the upper, middle and lower classes.
1856: Burberry, maker of iconic plaid scarves, was founded.
1900: Isadora Duncan, considered by many to be the mother of modern dance, popularizes long flowing scarves. Ironically, Duncan died as the result of a freak accident during which her long scarf was caught in the wheel of an automobile.
1914: The knitting of scarves became a patriotic war-time duty in the United States.
1930: Fur scarves were at the height of fashion in France.
1970: It was popular to wear scarves as a headband across the forehead or wrapped about the waist and chest as shirt.
2004: France passed a law that bans the wearing of Muslim head scarves in public schools.
2014 Scarf Festival
In Australia, we celebrate the scarf apparel with a yearly themed "Scarf Festival". In 2003, the scarf was first honoured with a festival that was held at Craft Victoria in Melbourne, Victoria, Australia. In 2008 the Scarf Festival moved to the National Wool Museum in Geelong, Victoria.
The National Wool Museum houses the timeless Australian story of wool, combined with exciting contemporary exhibitions, presented in an historic bluestone woolstore in Geelong's vibrant waterfront precinct.
Photograph Courtesy National Wool Museum.
This is the sixth year that the National Wool Museum has hosted the Scarf Festival. “Divinely wicked or devilishly good?” was the theme of this year’s Scarf Festival which attracted a record 329 scarves from across Australia and overseas. The National Wool Museum invited participants to explore and interpret the theme “Divinely wicked or devilishly good?” according to the suggested lines of thought:
(i) Will they be tempted by good or bad, or naughty or nice?
(ii) What happens when darkness meets the light?
(iii) What does it mean when the lines blur and the balance is broken?
(iv) How will they say it in a scarf?
The festival officially opened on June 5th, when a crowd of more than 150 gathered at the museum for the annual fashion show and award-winner announcements. The festival attracts entrants from some of Australia’s and the world’s well-known crafters and artists. Techniques include knitted, crocheted, woven, sewn, appliqued, dyed, printed and embellished pieces to name a few. The weirdest, wonderful, avant-garde and innovative creations are on show. Many of the scarves are available for purchase.
There were 12 Categories and winners in those categories were:
Scarf of the Year 2014: Margaret Drayton.
Thematic: Janette Wotherspoon.
Use of Colour: Di Liddelow.
Menswear: Christine Durbridge.
100% Wool: Kathleen Williams.
Sustainable Practices: Jo Bagge.
Woven: Kim Doherty.
Knitted: Kitty Morris.
Crocheted: Margaret Drayton also Scarf of the Year 2014 Winner.
Felted: Beverley Cox.
Primary School: Emily Bagge.
Secondary School: Lily Geyle.
Visitors could vote for their favorite scarf for the People's Choice Award at the festival. Craft demonstrations were held daily from 10:00am to 4:00pm during the course of the festival. The festival runs until September 7th 2014 at the National Wool Museum, 26 Moorabool Street, Geelong, Victoria, Australia.
My Scaves@2014 Scarf Festival - "Urban Artscape" Pashmina’s
My entries for this year’s Scarf Festival were based on the Thematic category which I interpreted as the "Urban Artscape".
The thematic elements in the works centred around the highly colorful and dynamic visual language of graffiti/street art, which abounds in our urban cityscapes. To some, this art form is considered as "nuisance" art. To me, street culture art encompasses the "devilishly good". By employing simplistic tools and mark making, there is a kindred spirit linking the art of the huntsman of prehistoric times to present day graffiti artists.
My first scarf was my Sampler. I then proceeded to create three large "Urban Artscape" pashmina scarves for the Festival based on the colors of the graffiti palette.
The scarves were hand dyed and hand printed and employed color washes, drawings and multiple mono print techniques using fabric pigments and permanent markers on viscose fabric. Each pashmina scarf measured 74 (width) x 195 (height) cm. Special care instructions were included with each scarf.
Excluding the Sampler, each scarf is available for purchase from the National Wool Museum during the festival which concludes on the 7th September 2014.
"Urban Artscape - Pashmina Sample" (full view).
"Urban Artscape - Pashmina Sample" (detail).
"Urban Artscape - Pashmina Sample" (close-up).
"Urban Artscape Pashmina 1" (full view).
"Urban Artscape Pashmina 1" (detailed view).
"Urban Artscape Pashmina 1" (close-up view).
"Urban Artscape Pashmina 2" (full view).
"Urban Artscape Pashmina 2" (detailed view).
"Urban Artscape Pashmina 2" (close-up view).
"Urban Artscape Pashmina 3" (full view).
"Urban Artscape Pashmina 2" (detailed view).
"Urban Artscape Pashmina 2" (close-up view).
Reference:
[1] https://www.scarves.net/blog/the-history-of-scarves-a-timeline/
Preamble
This is the twenty-ninth post in the "Art Resource" series, specifically aimed to construct an appropriate knowledge base in order to develop an artistic voice in ArtCloth.
Other posts in this series are:
Glossary of Cultural and Architectural Terms
Units Used in Dyeing and Printing of Fabrics
Occupational, Health & Safety
A Brief History of Color
The Nature of Color
Psychology of Color
Color Schemes
The Naming of Colors
The Munsell Color Classification System
Methuen Color Index and Classification System
The CIE System
Pantone - A Modern Color Classification System
Optical Properties of Fiber Materials
General Properties of Fiber Polymers and Fibers - Part I
General Properties of Fiber Polymers and Fibers - Part II
General Properties of Fiber Polymers and Fibers - Part III
General Properties of Fiber Polymers and Fibers - Part IV
General Properties of Fiber Polymers and Fibers - Part V
Protein Fibers - Wool
Protein Fibers - Speciality Hair Fibers
Protein Fibers - Silk
Protein Fibers - Wool versus Silk
Timelines of Fabrics, Dyes and Other Stuff
Cellulosic Fibers (Natural) - Cotton
Cellulosic Fibers (Natural) - Linen
Other Natural Cellulosic Fibers
General Overview of Man-Made Fibers
Man-Made Cellulosic Fibers - Viscose
Man-Made Cellulosic Fibers - Esters
Man-Made Synthetic Fibers - Nylon
Man-Made Synthetic Fibers - Polyester
Man-Made Synthetic Fibers - Acrylic and Modacrylic
Man-Made Synthetic Fibers - Olefins
Man-Made Synthetic Fibers - Elastomers
Man-Made Synthetic Fibers - Mineral Fibers
Man Made Fibers - Other Textile Fibers
Fiber Blends
From Fiber to Yarn: Overview - Part I
From Fiber to Yarn: Overview - Part II
Melt-Spun Fibers
Characteristics of Filament Yarn
Yarn Classification
Direct Spun Yarns
Textured Filament Yarns
Fabric Construction - Felt
Fabric Construction - Nonwoven fabrics
A Fashion Data Base
Fabric Construction - Leather
Fabric Construction - Films
Glossary of Colors, Dyes, Inks, Pigments and Resins
Fabric Construction – Foams and Poromeric Material
Knitting
Hosiery
Glossary of Fabrics, Fibers, Finishes, Garments and Yarns
Weaving and the Loom
Similarities and Differences in Woven Fabrics
The Three Basic Weaves - Plain Weave (Part I)
The Three Basic Weaves - Plain Weave (Part II)
The Three Basic Weaves - Twill Weave
The Three Basic Weaves - Satin Weave
Figured Weaves - Leno Weave
Figured Weaves – Piqué Weave
Figured Fabrics
Glossary of Art, Artists, Art Motifs and Art Movements
Crêpe Fabrics
Crêpe Effect Fabrics
Pile Fabrics - General
Woven Pile Fabrics
Chenille Yarn and Tufted Pile Fabrics
Knit-Pile Fabrics
Flocked Pile Fabrics and Other Pile Construction Processes
Glossary of Paper, Photography, Printing, Prints and Publication Terms
Napped Fabrics – Part I
Napped Fabrics – Part II
Double Cloth
Multicomponent Fabrics
Knit-Sew or Stitch Through Fabrics
Finishes - Overview
Finishes - Initial Fabric Cleaning
Mechanical Finishes - Part I
Mechanical Finishes - Part II
Additive Finishes
Chemical Finishes - Bleaching
Glossary of Scientific Terms
Chemical Finishes - Acid Finishes
Finishes: Mercerization
Finishes: Waterproof and Water-Repellent Fabrics
Finishes: Flame-Proofed Fabrics
Finishes to Prevent Attack by Insects and Micro-Organisms
Other Finishes
Shrinkage - Part I
Shrinkage - Part II
Progressive Shrinkage and Methods of Control
Durable Press and Wash-and-Wear Finishes - Part I
Durable Press and Wash-and-Wear Finishes - Part II
Durable Press and Wash-and-Wear Finishes - Part III
Durable Press and Wash-and-Wear Finishes - Part IV
Durable Press and Wash-and-Wear Finishes - Part V
The General Theory of Dyeing – Part I
The General Theory Of Dyeing - Part II
Natural Dyes
Natural Dyes - Indigo
Mordant Dyes
Premetallized Dyes
Azoic Dyes
Basic Dyes
Acid Dyes
Disperse Dyes
Direct Dyes
Reactive Dyes
Sulfur Dyes
Blends – Fibers and Direct Dyeing
The General Theory of Printing
There are currently eight data bases on this blogspot, namely, the Glossary of Cultural and Architectural Terms, Timelines of Fabrics, Dyes and Other Stuff, A Fashion Data Base, the Glossary of Colors, Dyes, Inks, Pigments and Resins, the Glossary of Fabrics, Fibers, Finishes, Garments and Yarns, Glossary of Art, Artists, Art Motifs and Art Movements, Glossary of Paper, Photography, Printing, Prints and Publication Terms and the Glossary of Scientific Terms, which has been updated to Version 3.5. All data bases will be updated from time-to-time in the future.
If you find any post on this blog site useful, you can save it or copy and paste it into your own "Word" document etc. for your future reference. For example, Safari allows you to save a post (e.g. click on "File", click on "Print" and release, click on "PDF" and then click on "Save As" and release - and a PDF should appear where you have stored it). Safari also allows you to mail a post to a friend (click on "File", and then point cursor to "Mail Contents On This Page" and release). Either way, this or other posts on this site may be a useful Art Resource for you.
The Art Resource series will be the first post in each calendar month. Remember - these Art Resource posts span information that will be useful for a home hobbyist to that required by a final year University Fine-Art student and so undoubtedly, some parts of any Art Resource post may appear far too technical for your needs (skip over those mind boggling parts) and in other parts, it may be too simplistic with respect to your level of knowledge (ditto the skip). The trade-off between these two extremes will mean that Art Resource posts will hopefully be useful in parts to most, but unfortunately may not be satisfying to all!
Introduction
Textile fibers, which are composed of ester-cellulose, are called acetate fibers. There are two types: acetate and triacetate. Since acetate fibers have very similar properties both will be considered in this post. Note: In chemistry an ester is designated the chemical formula of "RCOOR", where "C" stands for carbon, "O" for oxygen and the "R" represents a large array of possible chemical groups. The term "acetate" is derive from "acet" and "ate", with the former derived from acetic acid (the acid of vinegar) and the latter donates a chemical salt. Acetate therefore means a salt of the acid of vinegar (i.e. a salt of acetic acid).
Acetate was developed about the same time as rayon and it was initially named as acetate rayon until 1952 when the US Federal Trade Commission recognised that it had a different chemical composition and properties to rayon and so ruled it could not be labeled as rayon. It therefore became known as acetate.
Acetate fibers are frequently used for women's dresses and exhibits their beauty in the form of women's formal wear, suits, coats or knitwear.
Triacetate was introduced in 1954 to serve slightly different needs, as it can be more easily washed. Anrnel is a trade name for triacetate.
This post will focus on acetate and triacetate fibers.
Source and Production
Acetate is made from cellulose materials such as wood pulp, but the manufacturing process results in a fiber that is chemically different from rayon. The cellulose is combined with acetic acid to create a new chemical compound, cellulose acetate. When a solvent, acetone, is added, the cellulose acetate dissolves into a honey-like consistency, ready to be forced through the holes of a tiny spinneret. Fine filaments are produced and twisted together, then wound on a bobbin in the form of a yarn.
To make a cellulose salt of acetic acid, such a salt is known as an ester in Organic Chemistry parlance. As a result, acetate and triacetate fibres are also referred to as cellulose esters or ester-cellulose fibers.
In the manufacture of ester-cellulose fibers, triacetate is produced first; it is known as the primary cellulose acetate fiber since it is fully acetylated which means that the six –OH groups of the cellobiose unit is converted to six –OCOCH3 (or acetate groups). The secondary cellulose acetate fiber (acetate) is hydrolyzed (reacted with water) so that theoretically only 2.3 or 2.4 acetyl or acetate groups per the glucose unit occur and so it is only partially acetylated.
Manufacture Of Acetate Polymers.
Courtesy of Japan Chemical Fiber Association.
Hence there are two types of acetate fibersthat can be formed in the above process:
(i) Acetate – a man-made natural polymer base that is called a secondary cellulose filament or staple fiber.
(ii) Triacetate – a man-made natural polymer base that is called a primary cellulose acetate filament or staple fiber.
World consumption of acetate cellulose fibers.
General Properties of Acetate and Triacetate Fibers
Fiber Density
The fiber density of these fibers is 1.32 g cm-3 which makes them medium weight fibers.
Micro Structure of Acetate and Triacetate Fibers
Both of the polymer systems of acetate and triacetate are linear, but the acetyl groups form bulky side groups. The acetate or secondary cellulose acetate polymer is about 160 nm long and about 2.3 nm thick, whereas the triacetate or primary cellulose acetate polymer is 240 nm long and about 2.6 nm thick.
Triacetate Fibers.
The important chemical groupings of the acetate polymers are the hydroxyl and acetate groups. The hydroxyl groups tend to indicate the existence of hydrogen bonding. However, these hydrogen bonds are few in number, and cannot be significant in maintaining the polymer system structure. The acetate groups of the secondary cellulose acetate polymers are essentially non-polar and out number the hydroxyl groups. This explains the lack of polarity of the acetate polymers. In regard to the triacetate, its polymers must have less than 8% of hydroxyl groups by definition, and so are too few in number to cause significant hydrogen bonding. The triacetate polymers also lack polarity for this reason. Hence both polymer systems rely on weak van der Waals forces for their structural integrity.
Acetylation changes hydrophilic cellulose into hydrophobic cellulose acetate.
Courtesy reference[1].
Macro Polymer Structure
The acetate polymer system is estimated to be 40% crystalline and 60% amorphous, whilst the triacetate is considered somewhat more crystalline. Both fibers are therefore very amorphous. The inter-polymer system is held together via hydrogen bonding, but mostly by weak van der Waals forces.
Since both types of acetate fibers have a polymer backbone of hexagonal units, their polymer systems could be visualized as a disarranged roll of chicken wire, the disarranged portions being the amorphous regions of the polymer systems, whilst the more orderly sections are the crystalline region of the polymer systems.
Burning Behaviour of Fibers
Continues to burn when removed from source of flame. May actually melt. Has a vinegar or acetic acid odour. Forms a hard, black plastic bead.
Fiber Strength
Not too strong, lacks abrasion resistance. For this reason, it is considered a beauty fiber. It is weaker wet than dry.
Moisture Absorbency
Absorbs less moisture than cellulose and protein fibers, but more than other thermoplastics. Because of this it is less likely to shrink and will dry more quickly. It is more resistant to soiling than cellulose and protein fibers, but less than other thermoplastics. Note: As a thermoplastic fiber it is sensitive to heat and so a hot iron will make the fabric soften, glaze, and even fuse and develop holes.
Effect of Acetone
Acetone is a solvent. Fingernail polish remover contains acetone and so will cause holes if spilled on triacetate and acetate fibers.
Color Fastness
Blue and green dyes on acetate fabrics will often fade due to the gases contained in air (primarily oxygen). Solution dyed fabrics are permanently colorfast.
Specific Triacetate Properties
Triacetate was primarily developed since it is stronger, can withstand higher temperatures and is less likely to shrink or stretch. Originally used for tennis dresses because it washes so well, stays white, and has a good body, it is well liked for knitted fabrics and for many other easy-care garments.
Care
Clothing made of acetate should be hand washed in luke warm water and rinsed well. Press on the wrong side while damp, with an iron at a low setting. Triacetate fabrics can be machine laundered and bleached if necessary. They can stand ironing at higher temperatures, preferably the wrong side. Many acetate garments should be dry cleaned.
Uses
Considered the beauty fiber, acetate has a pleasing hand and drape so it is often used for evening gowns and party frocks.
Triacetate is especially popular for jersey dresses, for travel wear, for summer pleated skirts, and other easy care needs.
Physical Properties
Tenacity
Both types of acetate fibers are weak due to the amorphous nature of their polymer systems, which limits the number of inter-polymer forces of attraction, which can occur. In addition, the predominant forces of attraction between polymer units are the weak van der Waals forces.
Acetate and triacetate become weaker when wet, which occurs because the water molecules enter the amorphous region of the polymer systems, thereby pushing the polymer units apart (i.e. swells the region), and so weakening the short range attraction of the weak van der Waals forces. This causes a loss in tenacity of the filament or staple fiber.
Elastic-Plastic Nature
Both acetate and triacetate are plastic because their amorphous regions being the dominant region, and because of weak van der Waals forces of attraction that occur between the polymer units. With such weak forces and because of their amorphous nature, slippage readily occurs, even under slight strains, causing these textile materials to readily distort and/or wrinkle.
Both acetate and triacetate fibers become more plastic when wet, since water entering the amorphous regions break a significant number of inter-polymer forces of attraction. The disruption of the van der Waals forces makes slippage even easier, under the slightest strain and so these fabrics are more likely to distort and/or wrinkle when wet or just damp.
Acetate filaments and staple fibers have the softest handling of all textiles in common use, which once again is due to the amorphous nature of the polymer system and the weak bonding between polymer units. When pressure, such as handling, is applied to these secondary cellulose textile materials, weak inter-polymer forces present little resistance, creating a soft handling sensation.
Triacetate fibers tend to have a stiffer handle, particularly after heat setting. It is considered that after heat setting, the triacetate fibers move closer together (see thermal properties below) providing a more rigid polymer structure due to van der Waals forces of attraction increasing significantly in magnitude (because of the smaller distance) and so, triacetates offer greater resistance to any pressure such as handling.
Hygroscopic Nature
Despite their amorphous nature, both acetate and triacetate fibers have only fair moisture absorbency, mainly due to their low polarity and so have little attraction to the water molecules, which are polar.
Acetate polymer contains about two hydroxyl groups per cellobiose unit, which makes these polymers slightly more polar and the filaments and staple fibers more absorbent than those of triacetate. The latter polymer system contains six ester groups, which impart very little, if any, polarity making it more hydrophobic (water hating) than acetate. The triacetate fiber can even become more water hating on heat setting. This is due to the triacetate polymers being forced closer together, thereby even reducing the size of the inter-polymer spaces. This restricts the entry of water molecules into the polymer system. In effect, heat setting reduces the moisture absorbency of the triacetate polymer system by about half.
Finally, the limited hygroscopic nature of both types of acetate fibers, makes them prone to a build up of static electricity in dry atmospheric conditions. The limited hygroscopic nature is due to the lack of polarity of the polymer systems, thereby preventing attraction of water molecules into the amorphous regions of the polymer system that would dissipate static electricity in dry climates.
Thermal Properties
The degree of polymerization of viscose is 175 and that of acetate and triacetate polymers is 130 and 225 respectively. This suggests that the acetate and triacetate polymer lengths are similar to viscose and much smaller than cotton. Hence the explanation of viscose poor heat resistance and conductivity can be applied to both the acetate and triacetate filaments, and to their staple fibers.
Both fibers are thermoplastic, which means that they may be shaped, set, creased or pleated by the application of heat. When acetate textile materials are heated, the inter-polymer forces of attraction between the polymer units are severed. This permits the acetate and triacetate polymers to assume the configuration required of them by the set that is being applied to their textile material. On cooling, the inter-polymer forms of attraction reform to hold the polymers in this new position.
Secondary cellulose acetate cannot be heat set satisfactorily because it has relatively few hydrogen bonds. When the system is subject to the slightest strains, the new inter-polymer configuration can be altered since this will severe the van der Waals forces of attraction between polymer units and so allow the acetate polymers to be readily displaced from their new configuration.
On the other hand, primary cellulose acetate will retain a heat set more satisfactorily than secondary cellulose acetate, even though the polymer system is held together by weak van der Waals forces. Generally, a setting of a thermoplastic polymer system is thought to occur via a rearrangement of the polymer system configuration, resulting in an increase in tenacity. However, in the case of triacetate, the increase in tenacity does not occur on heat setting. A rearrangement of the triacetate polymers does occur, since the triacetate material will display a stiffer more paper-like handle, after heat setting. In addition, heat setting reduces the moisture absorbency by about half.
The handle observation is consistent with the process of recrystallization, which implies that on heating, the polymer units of triacetate get closer together increasing the strength of the van der Waals forces of attraction between the polymer units, and on cooling this renewed crystallization allows for resistance to straining against the new set positions of their filaments and staple fibers, thereby giving the fabric a more paper-like handle. In addition, the triacetate polymer system is thought to contract, and so reduce in size the inter-polymer spaces, and so prevent water uptake (i.e. causing a significant reduction in moisture absorbency).
Chemical Properties
Effect Of Acids
As both of the acetate polymers have essentially a cellulose backbone, acids hydrolyze them, causing polymer degradation, and resulting in weakening and eventual destruction of textile materials.
The triacetate polymer system is somewhat more crystalline than that of acetate, and so textile materials of triacetate will be more resist to acid degradation.
Effect Of Alkalis
The celloluse backbone makes acetate and triacetate polymers more resistant to alkalis rather than to acids. However, the acetyl, acetate or ester side groups will be hydrolyzed or saponified (i.e. hydrolysis of an ester to an alcohol) on exposure to alkaline conditions. The effect of this is the conversion of the acetate groups to hydroxyl groups as are found in the original cellulose polymer.
Such alkaline hydrolysis occurs in the first instance on the surface of the filaments or staple fibers, resulting in a yellowing of white or dulling of color acetate and triacetate textile materials.
Effect Of Bleaches
Bleaches in general have the same effect on acetate fibers as they do on cotton fibers.
Effect of Sunlight and Weather
The relatively lack of polarity of the acetate and triacetate polymers is one of the factors contributing to fair to good sunlight and weather resistance. The lack of polarity protects the polymers from destruction since in general most of the pollutants and degrading agents possess polarity.
Finally, acetate textile materials have fair to good sunlight resistance because of their electronic configurations are stable and so resist the degrading effect of UV light.
Color-Fastness
The acetate fibers are not easy fibers to dye or print, due to the relative lack of polarity. Disperse dyes have been specifically developed for this purpose (see below). Using these dyes, both polymer systems show good light- and wash-fastness.
Disperse Dyes
Disperse dyes are relatively non-polar and so they are also known as non-ionic dyes. Their lack of polarity makes them compatible with the relatively non-polar acetate and triacetate polymer systems.
The fair to good light-fastness of disperse dyed and printed acetate and triacetate textile material is due to their stable electronic arrangement of the chromophores of the disperse dye molecules, which can withstand the degrading effects of UV light.
The good wash-fastness of disperse dyed and printed acetate and triacetate textile materials is due to the non-polar nature of the disperse dye molecules, which are insoluble in water. These dye molecules are water hating and so the weak van der Waals forces of attraction together with the dye molecule entanglement in the amorphous regions of the polymer systems all contribute, albeit in different order of significance, in preventing water to rinse out of the disperse dye molecule from the acetate textile materials.
Acetate Satin Fabric.
Note: It tends to look like silk.
References:
[1] A Fritz and J. Cant, Consumer Textiles, Oxford University Press, Melbourne (1986).
[2] E.P.G. Gohl and L.D. Vilensky, Textile Science, Longman Cheshire, Melbourne (1989).
[3] E. J. Gawne, Fabrics for Clothing, 3rd Edition, Chas. A. Bennett Co., Peoria (1973).